Professor Karst Hoogsteen’s work and papers on the nucleoside published in Acta. Crystallagraphica between 1959 and 1963 are formidable pieces of work that have been ‘long buried’ precisely because they contradicted Crick and Watson’s base pairing solution. Interestingly his work directly supports the geometric pentagonal pairings that I propose - in every instance and diagram of his paper he illustrates the pentagonal part of the purine hydrogen-bonding with the pyrimidine and he states categorically that, given free will, the molecules refused to pair in the fashion proposed by Crick and Watson.
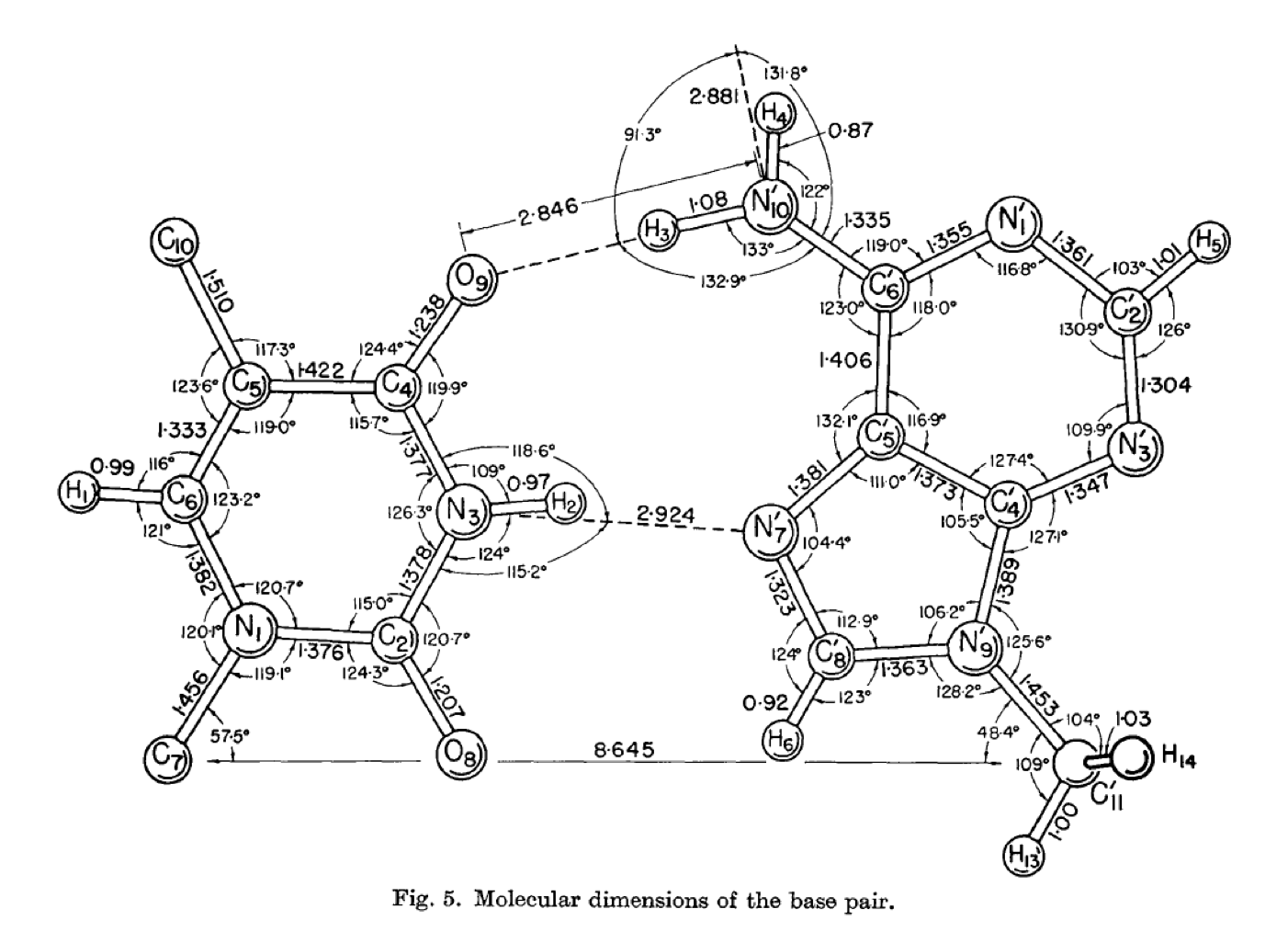
Karst Hoogsteen (1963) Acta. Cryst. vol 16, 907-916
His large and beautifully illustrated diagram on page 913 of his 1963 paper (above) actually shows a collapsed version of an A/T keto base pair illustrated in my publication. The same diagram is shown with geometric over-drawing (below).
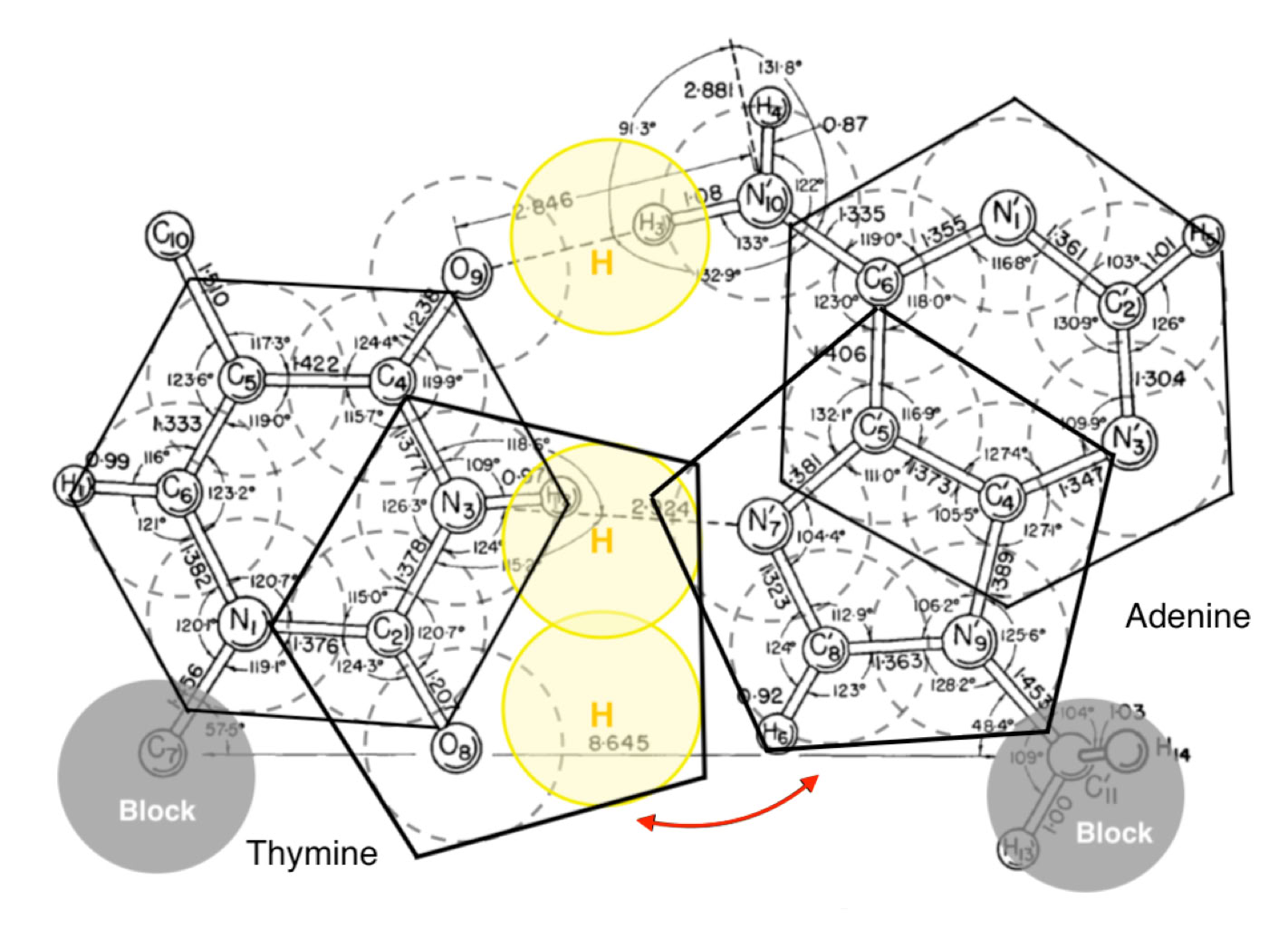
Geometric overdrawing of the Karst Hoogsteen base pair
Interestingly he states that he did not have as much success with G/C pairings as with A/T and this I believe likely to be a result of the blocks that he placed on the respective N1 pyrimidine and N9 purine positions in accordance with Crick and Watson’s proposal. I mention in my publication that the geometric proposal would require N9 of guanine to be available to form hydrogen bonds, as was indeed the case prior to 1953 and Crick & Watson fixing the sugar position. It would be interesting to repeat Hoogsteen’s research, especially with respect to Guanine and Cytosine, without blocking any of the potential hydrogen atoms.
As a point of note: I do appreciate that bond lengths and angles will not adhere precisely to that of the mathematical structure. The molecular pentamer is unlikely to fall within the exact measurements of a compass derived pentagon. It is the general principal of helical generation that I am concerned with and I would therefore expect a relative degree of variation - perhaps by as much as 9.5 to 10.5 bases per helical turn but maybe more.
It is also worth mentioning that structures and patterns that make use of the pentagon destroy geometric congruence, a necessary means by which energy is able to dissipate. The triangle, cube, hexagon, octagon etc, being congruent, will have a natural tendency to dissipate energy. This is potentially significant because congruence allows energy to flow seamlessly, whilst incongruence tends to focus energy. Johannes Kepler mentions that the pentagon, being incongruent, is deemed unsociable by reason of the fact that it ‘focuses' rather than ‘dissipates' [energy] (Johannes Kepler - Harmony of the World, Book II, prop IX). The upshot of this point being that it is possible the hydrogen bonds formed utilising the pentamer section of the purine have a stronger energy interaction with the pyrimidines than scientific understanding presently allows for.